
(Hydrogen wouldn’t work because it can form only one bond.)īut atoms that typically form only two chemical bonds, such as oxygen, don’t often make long, polymer-like chains. In theory, any atom that can form two chemical bonds can make a chain it’s like needing two hands to link with other people to make a circle. Zerbor/iStockphotoĪ chemical bond is what holds atoms together in a molecule and some crystals. It’s made of two carbon atoms, three hydrogens and one chlorine atom. For example, polyvinyl chloride (PVC) is made by linking long chains of monomers (shown in the bracket). Polymers are made by chemically linking up many copies of simpler groups called monomers. The longer the crosslinks, however, the more flexible a material becomes. They also make the polymer harder and more difficult to melt. They tend to strengthen a material (such as a plastic) made from this polymer. (Think of this as looking like a rung that stretches between the legs of a ladder.) Chemists refer to these ties as crosslinks. Sometimes pendant groups, instead of hanging loose from one polymer chain, actually connect two chains together. Because they are exposed to the surroundings more than are the atoms that make up the chain itself, these “charms” often determine how a polymer interacts with itself and other things in the environment. That’s because these groups hang off the main chain of the polymer just as individual charms hang off the chain of a charm bracelet. Others may be more complex and referred to as pendant groups. One of these attachments may be as simple as a single atom. Some may also have secondary parts that dangle from some (or all) of the chain’s links. All start with a basic chain of chemically bonded links. Polymer structures can have two different components. More often, chemists create polymers from compounds made in the lab. Or they can use amino acids to build artificial proteins unlike any made by Mother Nature. They may build artificial polymers from natural ingredients. In the lab, chemists have many options as they design and construct polymers. Although scientists have discovered some 500 different amino acids, animals and plants use only 20 of them to construct their proteins. Living things build proteins - a particular type of polymer - from monomers called amino acids. Others include the starch molecules made by plants. Cellulose, the main component of wood and paper, also is a natural polymer. Wool, cotton and silk are natural polymer-based materials that have been used since ancient times. The reason: They have a greater surface area, which makes them want to stick to neighboring molecules. Also, the longer a polymer chain, the higher its viscosity (or resistance to flow as a liquid). And, in general, longer polymers will give the materials made from them a higher melting and boiling temperature. The longer a polymer chain, the heavier it will be. Polymer chains can include hundreds of thousands of atoms - even millions. They are so big, in fact, that scientists classify them as macromolecules. Regardless of their shape, the molecules are very big. In some cases, polymers form branching networks rather than single chains. But in proteins, DNA and other natural polymers, links in the chain often differ from their neighbors.ĭNA, life’s storehouse of genetic information, is a lengthy molecule made from a series of smaller, repeating chemical units. In an artificial polymer, each of the chain’s links will often be identical to its neighbors. Think of a polymer as a chain, with each of its links a monomer. Those monomers can be simple - just an atom or two or three - or they might be complicated ring-shaped structures containing a dozen or more atoms. The word polymer comes from the Greek words for “many parts.” Each of those parts is scientists call a monomer (which in Greek means “one part”). Even the DNA in your cells is a polymer.īy definition, polymers are large molecules made by bonding (chemically linking) a series of building blocks. Consider keratin (KAIR-uh-tin), the stuff your hair and nails are made from. Many proteins in your body are polymers, too. The rubber in the tires on the family car. The nylon and polyester in your jacket or sneakers. The silicone rubber tips on your phone’s earbuds. 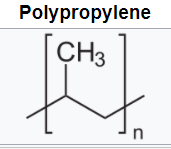
\): Polyisoprene.Polymers are everywhere.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
